57.5% achieved remission
by week 10*
Flow FL-100 has demonstrated clinically meaningful reductions in depressive symptoms across the largest controlled trial1 of its kind and real-world clinical practice2, including outcomes from more than 55,000 patients treated in routine care settings.
Potential amplification when used as an adjunct†1
Results at 10 weeks from a multisite, double-blind, placebo-controlled randomised superiority trial demonstrate that Flow FL-100 is effective when used both as a standalone treatment and as an adjunct to antidepressant therapy.
Remission rates when used as an adjunct and standalone at Week 10†1
.png?width=2000&height=1384&name=Group%2017537%20(1).png)
Week 20 open-label phase response rates1
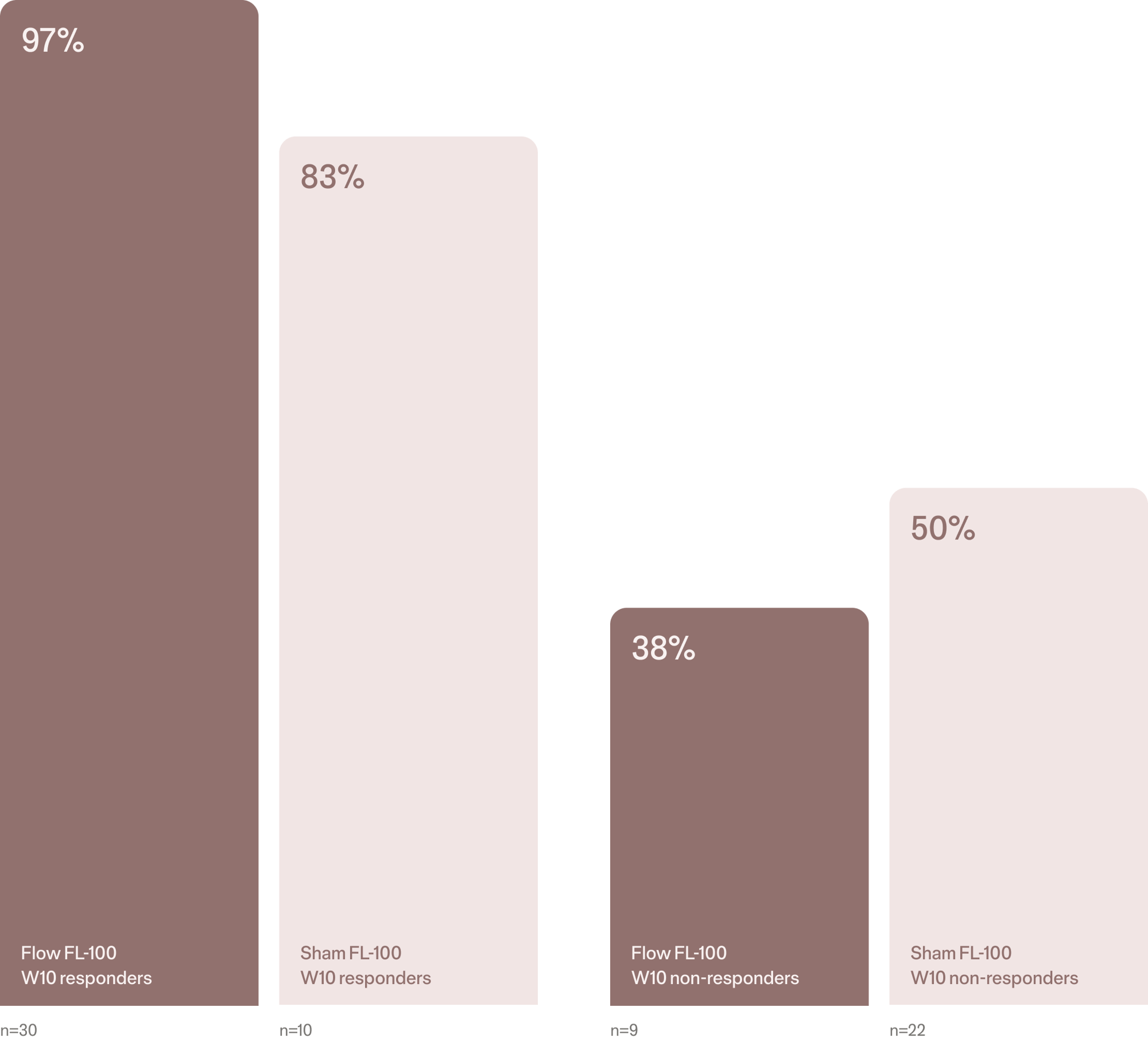
Response beyond 10 weeks
Follow-up data indicates that many patients who responded early to Flow FL-100 maintained their clinical improvement at Week 20.
Importantly, a subset of patients who had not achieved response by Week 10 experienced delayed improvements with continued treatment. These findings suggest that benefits may continue to emerge beyond the initial treatment period, highlighting the value of ongoing monitoring and structured treatment.
Week 20 open-label phase response rates1
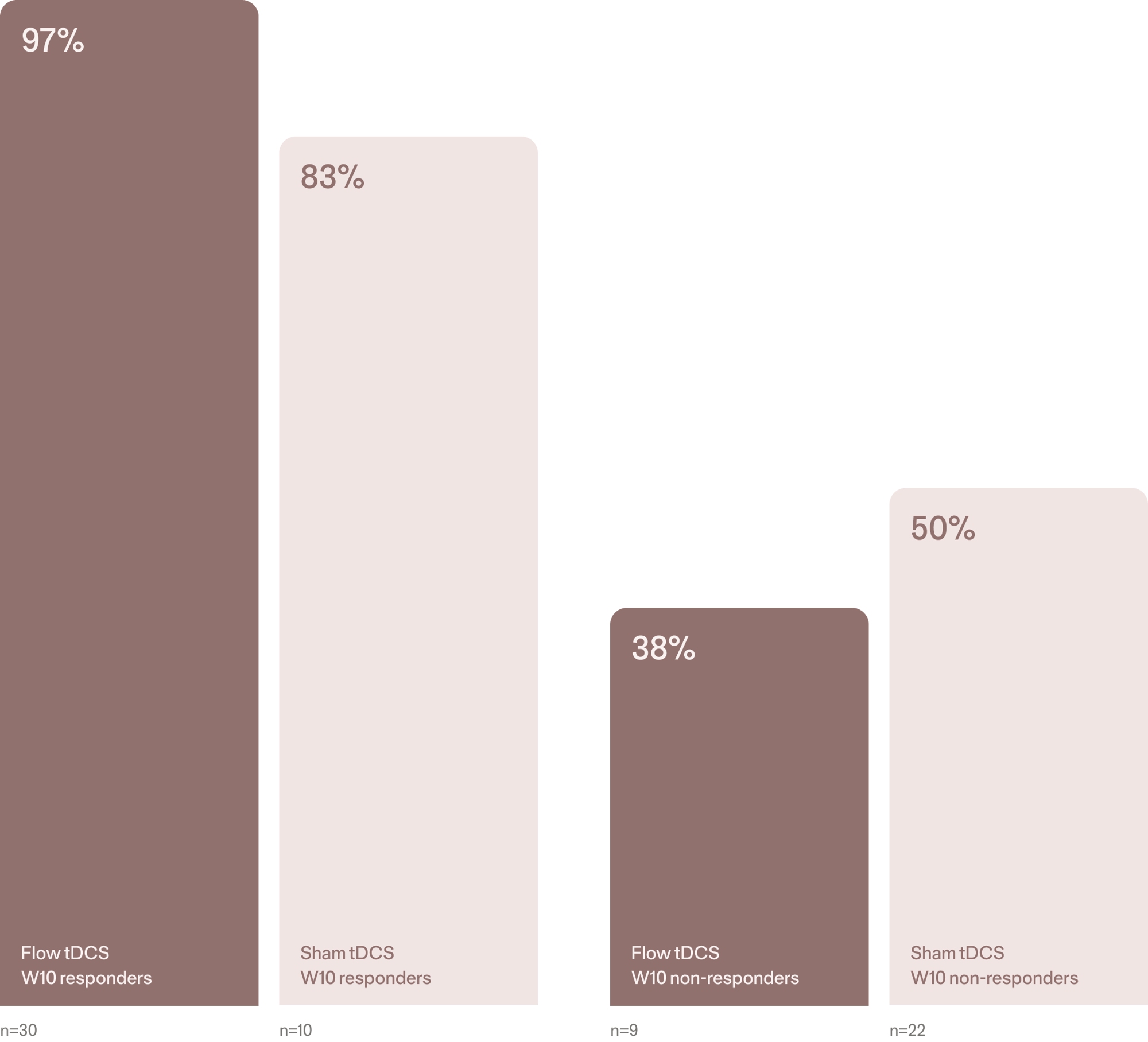
Flow is supported by a randomised controlled trial published in Nature Medicine and is already used in routine clinical care across the UK and EU, with more than 55,000 patients treated in real-world settings to date2.

Evidence beyond the trial setting
55,000+ users to date
30+ countries
Over 2 million Stimulations completed
In addition to controlled trial data, Flow FL-100 has been used in routine clinical practice across the UK and EU2. Real-world outcomes from tens of thousands of patients provide further insight into adherence, durability of response, and clinical utility outside of research settings.
.png?width=2000&height=1348&name=Frame%206%20(1).png)
Addressing risk in real-world clinical settings‡
In a real-world crisis care evaluation3, Flow FL-100 use was associated with up to a 75% reduction in reported suicidal ideation and avoided psychiatric hospital admissions, suggesting potential value as a supportive intervention for patients with acute depressive symptoms.
Change in distribution of Depression Severity after 3 weeks3‡
.png?width=2000&height=1298&name=Group%2017551%20(1).png)
Sleep quality remission rates among patients adherent to the tDCS protocol4
.png?width=1580&height=1394&name=Group%2017552%20(1).png)
Depression-related sleep symptoms in real-world use
In a retrospective analysis of over 6,000 patients treated with Flow FL-100 for depression, adherent patients showed notable improvements in insomnia, a common symptom of depression, with remission observed in 36% after 1 week and 63% by 10 weeks4.
Sleep quality remission rates among patients adherent to the tDCS protocol4
.png?width=1580&height=1394&name=Group%2017552%20(1).png)
Interested in prescribing Flow FL-100 to your patients? Be first to receive updates as we roll out treatment availability in the US.
*Remission is defined as a score of <10 on MADRS. MADRS remission rate was a secondary endpoint of the study. The primary endpoint of the study reviewed between-group mean difference in HDRS-17 scores and was –2.3 points in favour of the active group (p=0.012).
†Comparison of monotherapy and adjunctive use of Flow FL-100 was a sub-analysis evaluation included as table 10 and 11 in the Empower Study.
‡Findings from this crisis care evaluation are based on outcomes assessed after 3 and 6 weeks of treatment. The FDA-pivotal clinical trial evaluated its primary endpoint at 10 weeks; this difference in timeframe reflects the design and operational context of the crisis care service evaluation.
Please see more information about Flow FL-100, including the FDA-approved labeling, available here.
References: 1. Woodham, R.D., Selvaraj, S., Lajmi, N. et al. Nat Med (2024). 2. Data on file. Flow Neuroscience Real-world reports. 2025. 3. Griffiths, C., Abdalla, S., Mcconnochie, M. and Jiang, H. (2024). Open Journal of Psychiatry, 14, 514-527. 4. Silva, K., Griffiths, C., Banazadeh, A. and Mu, M. (2025). Open Journal of Psychiatry, 15, 109-121.
Indication
Flow FL-100 is intended for the treatment of moderate to severe major depressive disorder (MDD) in the current episode, either as monotherapy or as an adjunctive treatment, in patients 18 years and older who are not considered treatment refractory to medication.
Safety information
The side effects of the Flow FL-100 reported in clinical studies were generally mild and transient. The main risks of the device are skin dryness, skin irritation or redness after prolonged use, transient headache, stinging, burning, or itching sensations at the stimulation site, and skin burns (typically in cases of electrode pad reuse or use of dried-out electrode pads which is not recommended).
